Transcatheter Mitral Valve Implantation (TMVI) Pipeline by Development Stages, Segments, Region and Countries, Regulatory Path and Key Companies
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Transcatheter Mitral Valve Implantation (TMVI) Pipeline Market Report Overview
The transcatheter heart valve is an artificial heart valve made up of natural tissue surrounded by a flexible mesh frame known as stent. All the components are placed in a long narrow tube known as a catheter which is then inserted into a large blood vessel in the groin area or through a small incision in chest.
The transcatheter mitral valve implantation (TMVI) pipeline market research report provides a comprehensive understanding of the pipeline products along with their comparative analysis at various stages of development. The report also includes information about the territories wherein the clinical trials are in progress, the regulatory paths followed by the trials, and the companies associated with the trials.
| Key Territories | · The US
· China · Europe · Canada · Israel |
| Key Regulatory Paths | · PMA
· NMPA · CE · MDL · BOPA · MDCO |
| Leading Companies | · 4C Medical Technologies Inc.
· Abbott Vascular Inc. · Annulon · Cardiovalve Ltd · Cephea Valve Technologies, Inc. |
| Enquire & Decide | Discover the perfect solution for your business needs. Enquire now and let us help you make an informed decision before making a purchase. |
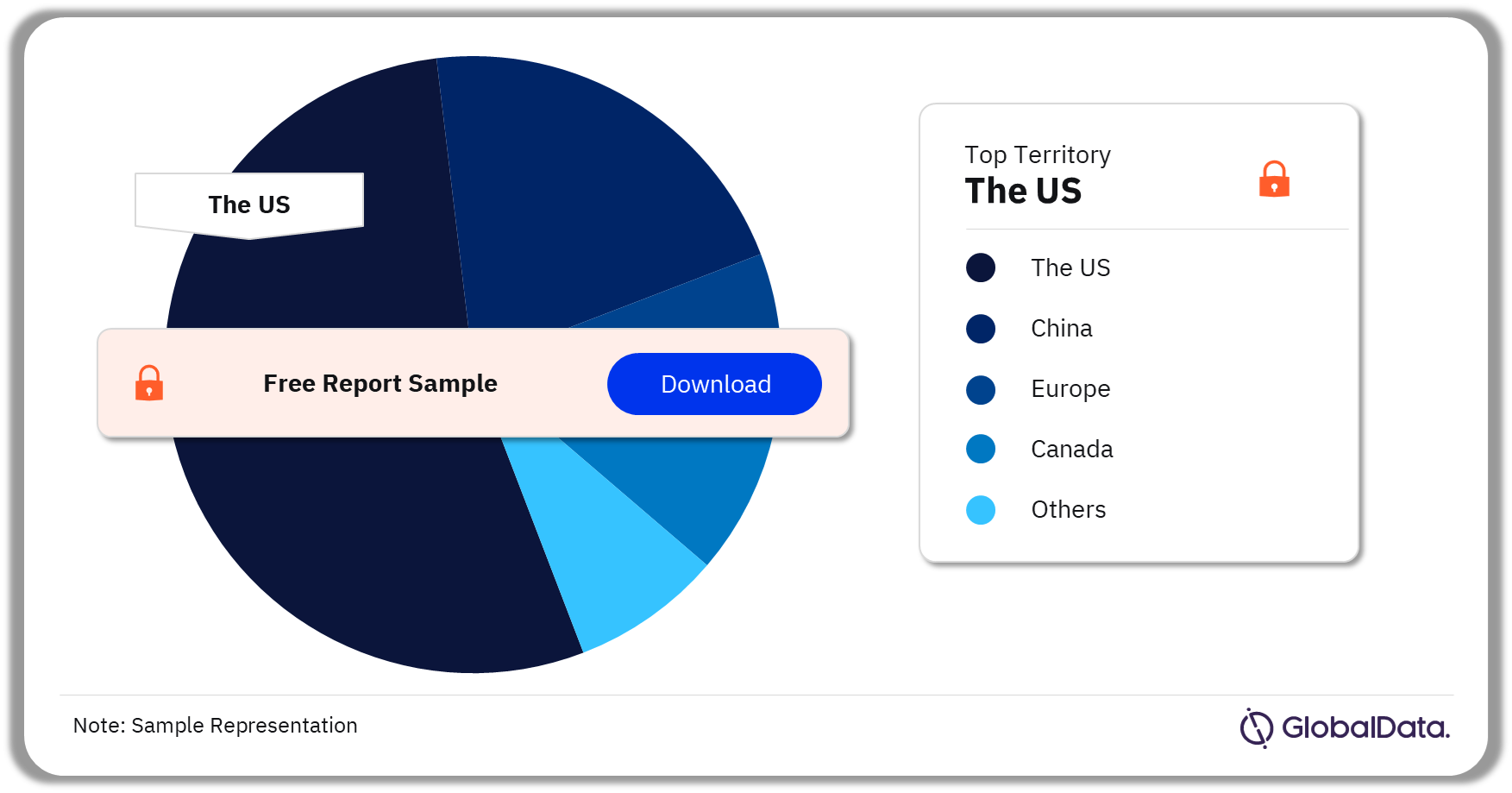
TMVI Pipeline Market Segmentation by Territories
A few of the key territories for the TMVI pipeline market are the US, China, Europe, Canada, and Israel. As of March 2024, the US accounted for the highest number of pipeline products.
TMVI Pipeline Market Analysis by Territories, 2024 (%)
Buy the Full Report for More Territory Insights into the TMVI Pipeline Market
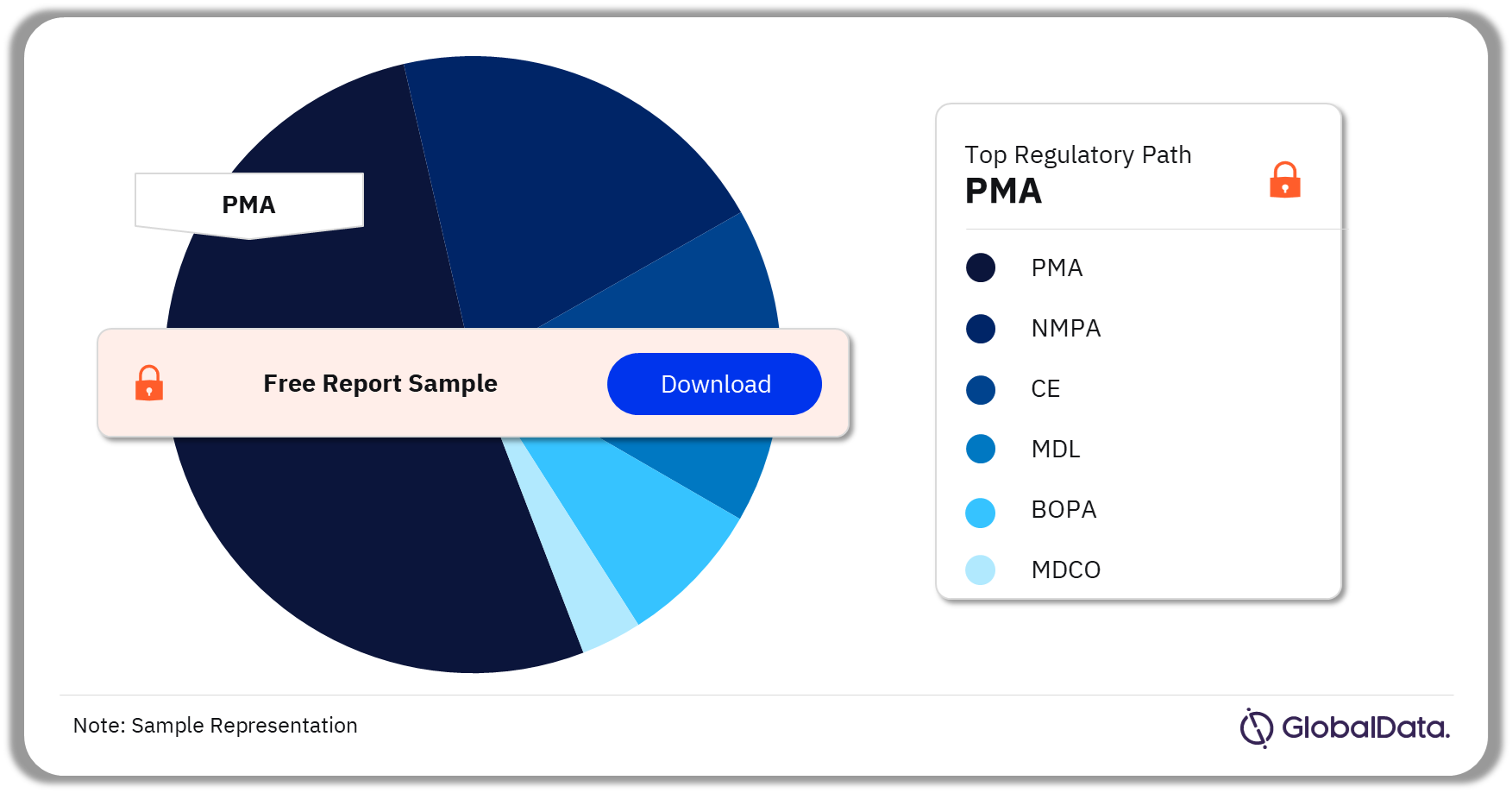
TMVI Pipeline Market Segmentation by Regulatory Paths
The key regulatory paths in the TMVI pipeline products market are PMA, NMPA, CE, MDL, BOPA, and MDCO. As of March 2024, PMA was the most followed pathway for pipeline products in the market.
TMVI Pipeline Market Analysis by Regulatory Paths, 2024 (%)
Buy the Full Report for More Regulatory Path Insights into the TMVI Pipeline Market
TMVI Pipeline Market – Competitive Landscape
A few of the key companies associated with the TMVI pipeline market are 4C Medical Technologies Inc., Abbott Vascular Inc., Annulon, Cardiovalve Ltd, and Cephea Valve Technologies, Inc., among others.
4C Medical: It is a medical device company developing minimally invasive therapies for structural heart disease. 4C Medical focuses initially on mitral regurgitation (MR) therapy and subsequently on tricuspid regurgitation therapy. The company’s implant is the first MR therapy with supra-annular positioning and atrial-only fixation.
Abbott Vascular Inc.: Abbott Vascular is headquartered in Santa Clara, California, the US. It is a medical device company that develops, manufactures, and markets cardiovascular devices. The company offers coronary intervention, peripheral intervention, and structural heart products. Abbott Vascular provides minimally invasive treatment for mitral regurgitation and other structural defects of the heart.
TMVI Pipeline Market Analysis by Companies, 2024
Buy the Full Report for More Company Insights into the TMVI Pipeline Market
Segments Covered in the Report
Transcatheter Mitral Valve Implantation (TMVI) Pipeline Market Territories Outlook
- The US
- China
- Europe
- Canada
- Israel
Transcatheter Mitral Valve Implantation (TMVI) Pipeline Market Regulatory Paths Outlook
- PMA
- NMPA
- CE
- MDL
- BOPA
- MDCO
Scope
This report provides:
- Extensive coverage of the transcatheter mitral valve implantation (TMVI) under development.
- Exhaustive list of major pipeline products and their details, including product description, licensing and collaboration details, and other developmental activities.
- Reviews of major players involved in the development of transcatheter mitral valve implantation (TMVI) and lists all their pipeline projects.
- Coverage of pipeline products based on various stages of development ranging from early development to approved/issued stage.
- Key clinical trial data of ongoing clinical trials specific to pipeline products.
- Recent developments in the segment/industry.
Reasons to Buy
- Formulate significant competitor information, analysis, and insights to improve R&D strategies.
- Identify emerging players with potentially strong product portfolios and create effective counter strategies to gain a competitive advantage.
- Identify and understand important and diverse types of transcatheter mitral valve implantation (TMVI) under development.
- Develop market-entry and market-expansion strategies.
- Plan mergers and acquisitions effectively by identifying major players with the most promising pipeline.
- Carry out an in-depth analysis of the product’s current stage of development, territory, and estimated launch date.
Abbott Vascular Inc
Annulon
Cardiovalve Ltd
Cephea Valve Technologies, Inc.
Colibri Heart Valve LLC
Direct Flow Medical Inc (Inactive)
Edwards Lifesciences Corp
Emory University
Foldax Inc
Jenscare Scientific Co Ltd
Jiangsu Zhenyi Medical Technology Co Ltd
Lepu Medical Technology (Beijing) Co Ltd
LivaNova PLC
Medtronic Plc
Micro Interventional Devices, Inc.
MicroPort CardioFlow Medtech Corp
MicroPort Scientific Corp
MitrAssist Ltd.
MValve Technologies Ltd.
Neovasc Inc
NewMed Medical Co Ltd
P+F Products + Features GmbH
Polares Medical SA
Shanghai Shenqi Medical Technology Co Ltd
Sino Medical Sciences Technology Inc
Symetis SA
Tioga Medical Inc
Trinity College Dublin
University College London
Valcare Medical
Venus Medtech Hangzhou Inc
Table of Contents
Table
Figures
Frequently asked questions
-
Which territory had the highest number of products in the TMVI pipeline market as of March 2024?
As of March 2024, the US accounted for the highest number of pipeline products in the TMVI market.
-
Which was the most followed regulatory pathway in the TMVI pipeline market as of March 2024?
As of March 2024, PMA was the most followed pathway for pipeline products in the TMVI market.
-
Which are the major companies operating in the TMVI pipeline market?
A few of the key companies associated with the TMVI pipeline market are 4C Medical Technologies Inc., Abbott Vascular Inc., Annulon, Cardiovalve Ltd, and Cephea Valve Technologies, Inc., among others.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Transcatheter Mitral Valve Implantation (TMVI) reports