Revision Hip Replacement Devices Pipeline Report including Stages of Development, Segments, Region and Countries, Regulatory Path and Key Companies, 2021
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Revision hip replacement is the replacement of the failed primary hip implants with a new implant. In revision hip replacement any of the damaged components (acetabular or femoral) of the hip joint are replaced. One unit refers to one implant set. GlobalData’s medical devices sector report provides comprehensive information about the revision of hip replacement devices pipeline products with a comparative analysis of the products at various stages of development and information about the clinical trials which are in progress.
What are the market dynamics in the revision hip replacement devices pipeline products market?
As of November 2021, 5 pipeline products were in the approval process and 3 were in the inactive stage. Only one pipelined product was in the early development stage. During the same period, based on pipeline products by stage of development, 4 products from JRI Orthopaedics Ltd were in the approval process. Japan is the leading country in the market based on pipeline products by territory, followed by the US and Europe.
Which are the key players in the revision hip replacement devices pipeline products market?
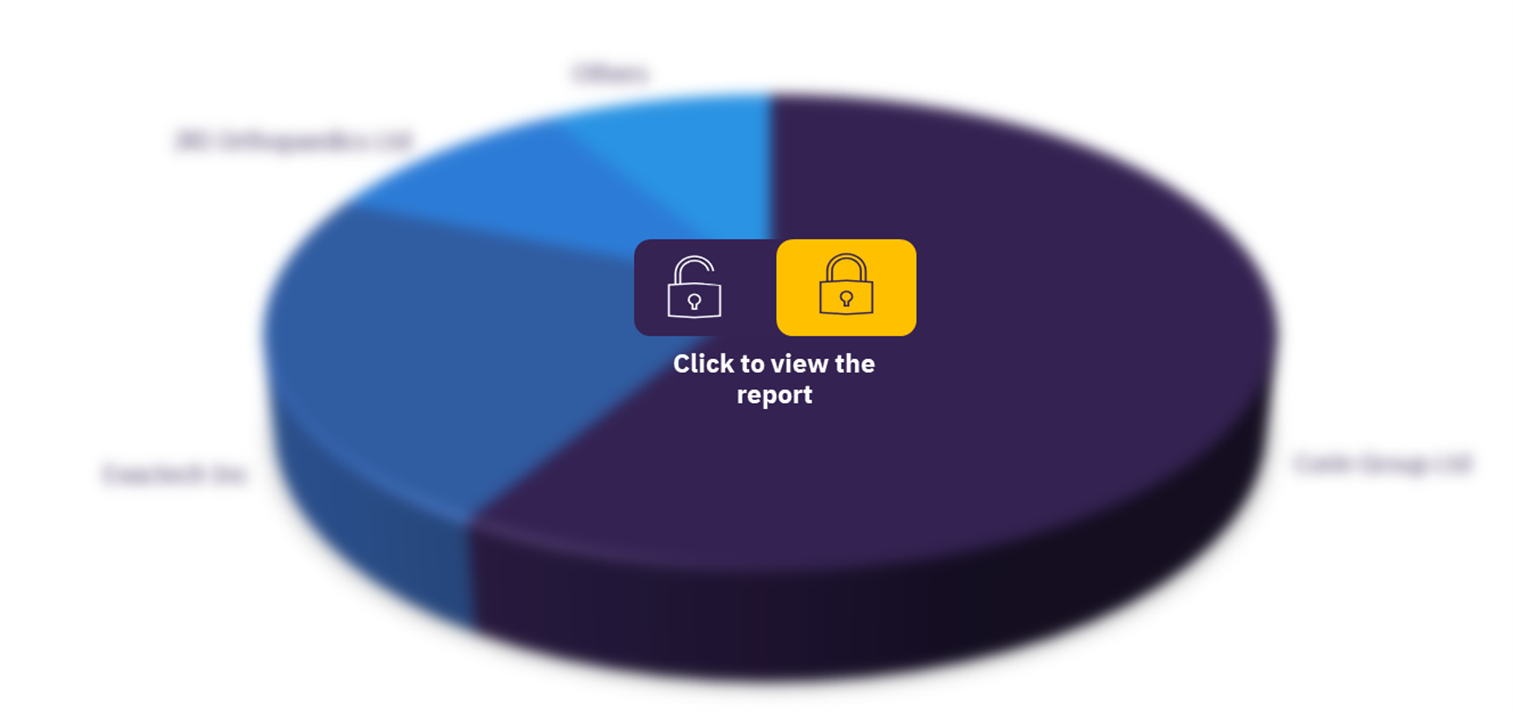
Key players in the market are Corin Group Ltd, Exactech Inc, JRI Orthopaedics Ltd, Limacorporate SpA, MicroPort Orthopedics Inc, and Pro-Dex Inc. JRI Orthopaedics Ltd is the leading company in the market based on the pipeline products in the approval process, followed by Corin Group Ltd.
Company overview:
Corin Group Ltd. overview: Corin Group Ltd (Corin Group), a subsidiary of Corin Orthopaedics Holdings Ltd, carries out the development, production, and distribution of orthopedic devices. The group offers orthopedic devices for knees, hips, extremities, trauma, and ligaments. Corin Group markets its products under brands including Rotaglide+, Trinity, MiniHip, MetaFix, TaperFit, Uniglide, LARS, Zenith, and others. The group offers solutions to hip, knee, ankle, and soft tissue. It operates in the Americas, Europe, Asia Pacific, the Middle East, and Africa. Corin Group is headquartered in Gloucestershire, England, the UK.
Exactech Inc.: Exactech Inc (Exactech) is a medical device company that offers orthopedic implant devices and related surgical instruments. The company’s products comprise hip implants, shoulder implants, knee implants, extremity implants, biologics and spine, and other products. It also provides biologic services. Exactech’s hip products comprise neck preserving stem, tapered wedge stem, element stem, tapered and splined stems, and others. The company caters to hospitals, surgeons, and other physicians and clinics. It operates in France, the UK, Taiwan, Germany, Japan, and others. Exactech is headquartered in Gainesville, Florida, the US.
JRI Orthopaedics Ltd: JRI Orthopaedics Ltd (JRI) a subsidiary of Orthopaedic Research UK (ORUK), is a manufacturer of orthopedic implants and surgical instruments. The company develops products for joint replacement, repair, and reconstruction. It offers products for shoulder and knee arthroplasty, besides customized shoulder and hip products and a range of orthobiologics. Its product portfolio comprises tribofit hip system, trekking knee arthroplasty system, Furlong evolution, and securus revision system, among others. The company works in partnership with clinical and non-clinical academics, and other commercial collaborators for the development of novel materials, advanced manufacturing processes, and regenerative medical products. JRI is headquartered in Sheffield, the UK.
Limacorporate SpA: Limacorporate SpA (Lima), a subsidiary of EQT Partners AB, is a medical device company that develops, manufactures, and commercializes reconstructive and fixation orthopedic solutions. The company’s product portfolio includes large joint primary and revision implants, extremities and fixation devices, patient-specific prosthesis (Promade) products for surgeons worldwide. Lima harnesses its proprietary trabecular titanium technology for additive implants manufacturing to develop prosthetic components that resemble natural bone. It also provides additive implants and biomaterials such as trabecular titanium and bioceramics. The company focus on niche segments of the orthopaedics market, including extremities implants for the shoulder and elbow, implants manufactured for revision, and other complex surgeries. It has operations with R&D and production facilities in Italy and San Marino. Lima is headquartered in Udine, Italy.
MicroPort Orthopedics Inc: MicroPort Orthopedics Inc (MicroPort Orthopedics), a subsidiary of MicroPort Scientific Corp is a medical device company that specializes in designing, manufacturing, and marketing of reconstructive joint devices and related services. The company manufactures orthopedic devices for the hip system, limb salvage, and knee systems. Its products include tissue-sparing surgical approaches, acetabular hip systems, primary and revision total knee systems, and other personalized solutions. MicroPort Orthopedics serves products and services for doctors, patients, and hospitals. The company has its presence in China, the Netherlands, Germany, the UK, Japan, Canada, France, Brazil, and other countries. MicroPort Orthopedics is headquartered in Arlington, Tennessee, the US.
Pro-Dex Inc.: Pro-Dex Inc (Pro-Dex) is a medical device company that manufactures and markets surgical devices. The company’s products include motion control, finished medical devices, micro air motors, plastic injection molding, dental products, and torque wrenches. Its dental products comprise DLS handpiece system, oral surgery handpiece, and prophy plus. Pro-Dex offers services such as design and technical drafting, electronics and software, mechanical engineering and design, precision machining and assembly, quality and compliance, testing, prototyping and validation, and precision injection molding. It serves healthcare, aerospace, automotive, industrial, communications, dental, research, robotics, and oil and gas industries. Pro-Dex is headquartered in Irvine, California, the US.
Revision hip replacement devices pipeline products market, by key players
To know more about key players, download a free report sample
Market report scope
| Key countries/regions | Japan, the US, and Europe |
| Key players | Corin Group Ltd, Exactech Inc, JRI Orthopaedics Ltd, Limacorporate SpA, MicroPort Orthopedics Inc, and Pro-Dex Inc |
Scope
- Extensive coverage of the Revision Hip Replacement Devices under development.
- The report reviews details of major pipeline products which include, product description, licensing and collaboration details, and other developmental activities.
- The report reviews the major players involved in the development of Revision Hip Replacement Devices and lists all their pipeline projects.
- The coverage of pipeline products is based on various stages of development ranging from Early Development to the Approved / Issued stage.
- The report provides key clinical trial data of ongoing trials specific to pipeline products.
- Recent developments in the segment/industry.
Reasons to Buy
The report enables you to:
- Formulate significant competitor information, analysis, and insights to improve R&D strategies.
- Identify emerging players with potentially strong product portfolios and create effective counter-strategies to gain a competitive advantage.
- Identify and understand important and diverse types of Revision Hip Replacement Devices under development.
- Develop market entry and market expansion strategies.
- Plan mergers and acquisitions effectively by identifying major players with the most promising pipeline.
- In-depth analysis of the product’s current stage of development, territory, and estimated launch date.
Exactech Inc
JRI Orthopaedics Ltd
Limacorporate SpA
MicroPort Orthopedics Inc
Pro-Dex Inc
Table of Contents
Table
Figures
Frequently asked questions
-
What are the key geographic regions in the revision hip replacement devices pipeline products market?
Japan is the leading country in the market based on pipeline products by territory, followed by the US and Europe.
-
Which are the key players in the revision hip replacement devices pipeline products market?
Key players in the market are Corin Group Ltd, Exactech Inc, JRI Orthopaedics Ltd, Limacorporate SpA, MicroPort Orthopedics Inc, and Pro-Dex Inc.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Orthopedic Devices reports