Transmucosal Drug Delivery – Pipeline Products by Stage of Development 21
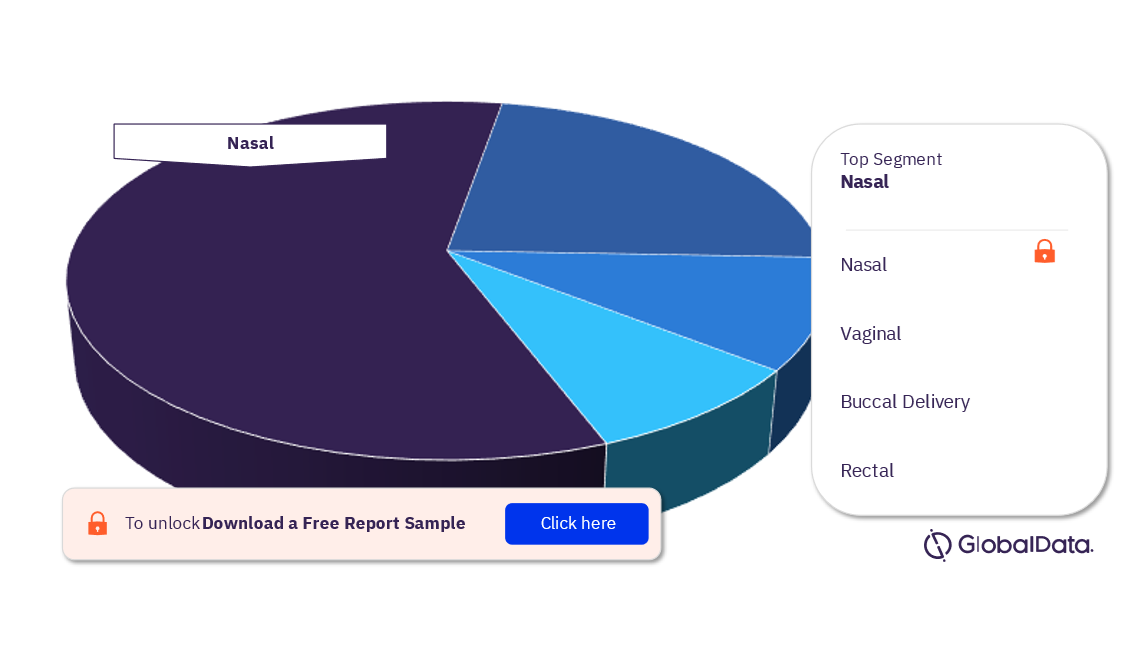
Transmucosal Drug Delivery – Pipeline Products by Segment 22
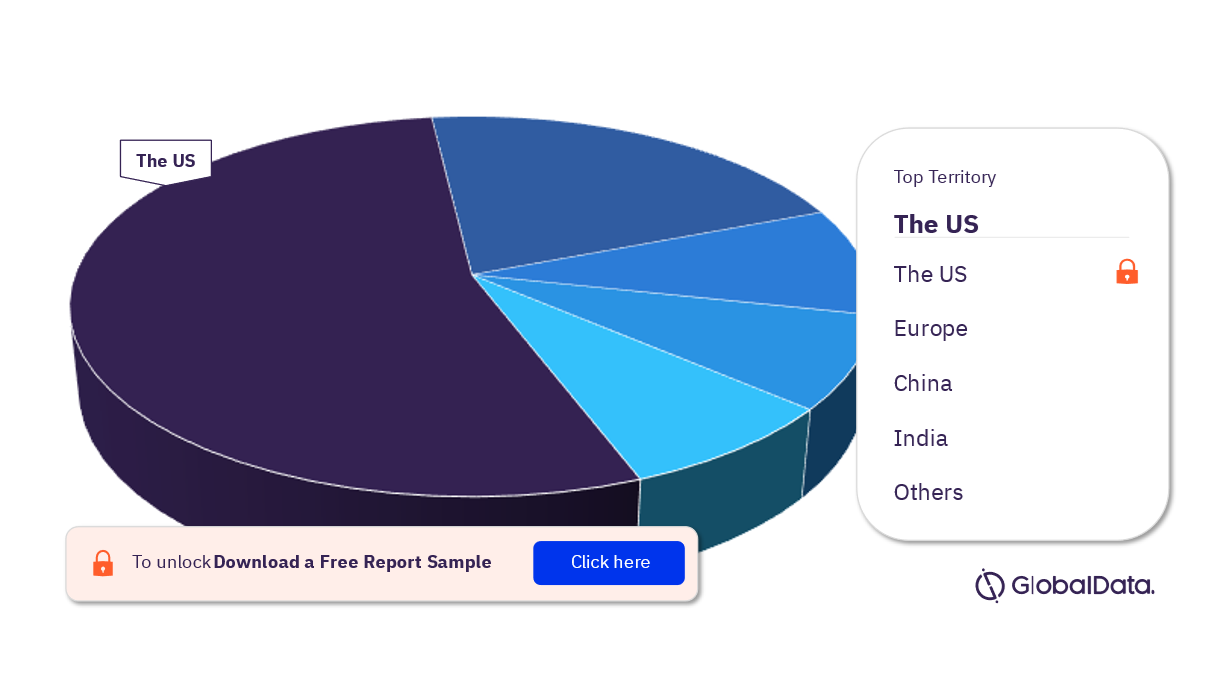
Transmucosal Drug Delivery – Pipeline Products by Territory 23
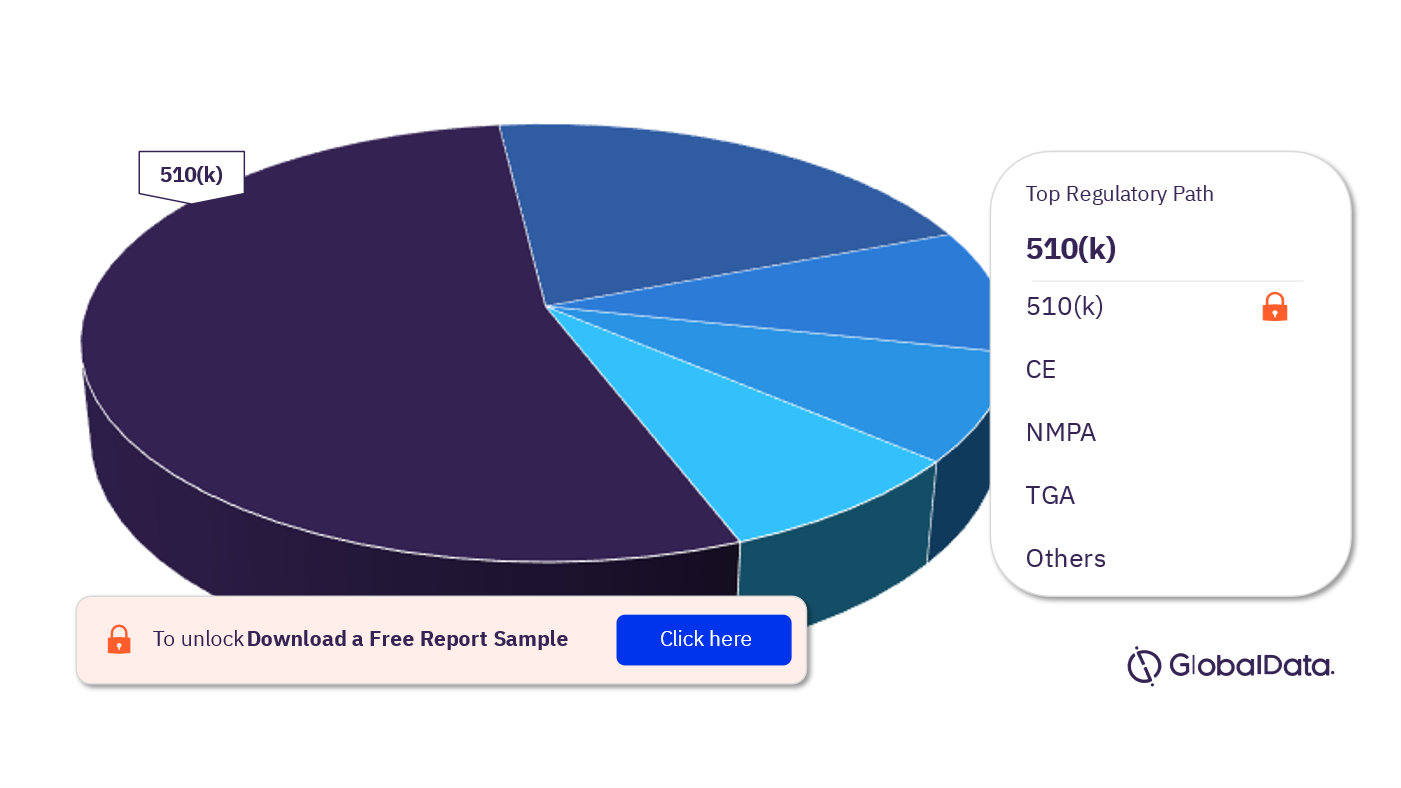
Transmucosal Drug Delivery – Pipeline Products by Regulatory Path 25
Transmucosal Drug Delivery – Pipeline Products by Estimated Approval Date 26
Transmucosal Drug Delivery – Ongoing Clinical Trials 27
Transmucosal Drug Delivery Companies – Pipeline Products by Stage of Development 28
Transmucosal Drug Delivery – Pipeline Products by Stage of Development 33
AcelRx Pharmaceuticals Inc Pipeline Products & Ongoing Clinical Trials Overview 37
Zalviso – Product Status 37
Zalviso – Product Description 38
AcelRx Pharmaceuticals Inc – Ongoing Clinical Trials Overview 39
Zalviso – Treatment of Acute Post-operative Pain in Patients Undergoing Laparoscopic Colon Resections 40
AFYX Therapeutics A/S Pipeline Products & Ongoing Clinical Trials Overview 41
Rivelin – CLO Patch – Product Status 41
Rivelin – CLO Patch – Product Description 41
Albert Einstein College of Medicine Pipeline Products & Ongoing Clinical Trials Overview 42
Drug-Impregnated Intravaginal Ring – Product Status 42
Drug-Impregnated Intravaginal Ring – Product Description 42
Allergan Ltd Pipeline Products & Ongoing Clinical Trials Overview 43
Vaginal Ring – Product Status 43
Vaginal Ring – Product Description 43
Altamira Medica AG Pipeline Products & Ongoing Clinical Trials Overview 44
Bentrio – Viral Infections – Product Status 44
Bentrio – Viral Infections – Product Description 44
Altamira Medica AG – Ongoing Clinical Trials Overview 45
Bentrio – Viral Infections – A Randomized, Placebo-controlled Clinical Study to Evaluate the Ability of Bentrio Nasal Spray in COVID-19 Patients: COVAMID 46
Bentrio – Viral Infections – Study Evaluating the Efficacy of Bentrio Nasal Spray for Protection Against Airborne Viruses and Allergens in India 46
Anelleo Inc Pipeline Products & Ongoing Clinical Trials Overview 47
AnelleO PRO – Product Status 47
AnelleO PRO – Product Description 47
Aquestive Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 48
Anaphylm – Product Status 48
Anaphylm – Product Description 48
Arovella Therapeutics Ltd Pipeline Products & Ongoing Clinical Trials Overview 49
ArTiMist – Product Status 49
ArTiMist – Product Description 49
ARS Pharmaceuticals, Inc. Pipeline Products & Ongoing Clinical Trials Overview 50
Neffy – Product Status 50
Neffy – Product Description 50
Atossa Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 51
AT-301 Nasal Spray – Product Status 51
AT-301 Nasal Spray – Product Description 51
Auris Medical AG Pipeline Products & Ongoing Clinical Trials Overview 52
AM-125 Betahistine Nasal Spray – Product Status 52
AM-125 Betahistine Nasal Spray – Product Description 52
AM-201 Betahistine Nasal Spray – Product Status 53
AM-201 Betahistine Nasal Spray – Product Description 53
Auritec Pharmaceuticals Inc Pipeline Products & Ongoing Clinical Trials Overview 54
BUP Pod-IVR – Product Status 54
BUP Pod-IVR – Product Description 55
NuvaRing – Product Status 55
NuvaRing – Product Description 55
pod-IVR – Product Status 56
pod-IVR – Product Description 56
Versa Intravaginal Ring – Acromegaly – Product Status 56
Versa Intravaginal Ring – Acromegaly – Product Description 57
Versa Intravaginal Ring – Alzheimer’s Disease – Product Status 57
Versa Intravaginal Ring – Alzheimer’s Disease – Product Description 57
Versa Intravaginal Ring – Breast Cancer – Product Status 58
Versa Intravaginal Ring – Breast Cancer – Product Description 58
Versa Intravaginal Ring – Endometriosis – Product Status 58
Versa Intravaginal Ring – Endometriosis – Product Description 59
Versa Intravaginal Ring – Herpes – Product Status 59
Versa Intravaginal Ring – Herpes – Product Description 59
Versa Intravaginal Ring – HIV Prophylaxis – Product Status 60
Versa Intravaginal Ring – HIV Prophylaxis – Product Description 60
Versa Intravaginal Ring – Pain And Addiction – Product Status 60
Versa Intravaginal Ring – Pain And Addiction – Product Description 61
Versa Intravaginal Ring – Parkinson’s Disease – Product Status 61
Versa Intravaginal Ring – Parkinson’s Disease – Product Description 61
Versa Intravaginal Ring – Type II Diabetes – Product Status 62
Versa Intravaginal Ring – Type II Diabetes – Product Description 62
Versa Intravaginal Ring – Uterine Fibroids – Product Status 62
Versa Intravaginal Ring – Uterine Fibroids – Product Description 63
BeFree! Pipeline Products & Ongoing Clinical Trials Overview 64
Vaginal Drug Delivery System – Product Status 64
Vaginal Drug Delivery System – Product Description 64
Bespak Europe Ltd Pipeline Products & Ongoing Clinical Trials Overview 65
NAS010 – Nasal Drug Delivery Device – Product Status 65
NAS010 – Nasal Drug Delivery Device – Product Description 65
NAS020 – Unidose Nasal Drug Delivery Device – Product Status 66
NAS020 – Unidose Nasal Drug Delivery Device – Product Description 66
NAS030 Nasal Device – Product Status 66
NAS030 Nasal Device – Product Description 66
Biorings LLC Pipeline Products & Ongoing Clinical Trials Overview 67
Vaginal Ring – Product Status 67
Vaginal Ring – Product Description 67
BioXcel Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 68
BXCL501 Sublingual Film – Product Status 68
BXCL501 Sublingual Film – Product Description 69
BioXcel Therapeutics Inc – Ongoing Clinical Trials Overview 70
BXCL501 Sublingual Film – A Multicenter, Randomized, Double-Blind, Placebo-Controlled, Efficacy and Safety Study of PRN Dosing of BXCL501 Over a 12 Week Treatment Period in Subjects with Agitation Associated with Dementia 71
BXCL501 Sublingual Film – A Study to Evaluate the BXCL501 for At-home Use in Patients with Bipolar Disorder or Schizophrenia 71
Birmingham Biotech Ltd Pipeline Products & Ongoing Clinical Trials Overview 72
BHM Anti-Viral Nasal Spray – Product Status 72
BHM Anti-Viral Nasal Spray – Product Description 72
Bryn Pharma LLC Pipeline Products & Ongoing Clinical Trials Overview 73
BRYN-NDS1C – Product Status 73
BRYN-NDS1C – Product Description 73
Callitas Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 74
CannaMint Strip – Product Status 74
CannaMint Strip – Product Description 74
Chulalongkorn University Pipeline Products & Ongoing Clinical Trials Overview 75
AntiCov Nasal Spray – Product Status 75
AntiCov Nasal Spray – Product Description 75
Combinent BioMedical Systems Inc Pipeline Products & Ongoing Clinical Trials Overview 76
Multi-Segmented Vaginal Ring – Product Status 76
Multi-Segmented Vaginal Ring – Product Description 76
Conrad Pipeline Products & Ongoing Clinical Trials Overview 77
Topical Dual-Compartment Insert – Product Status 77
Topical Dual-Compartment Insert – Product Description 77
Triple Protection Vaginal Ring – Product Status 78
Triple Protection Vaginal Ring – Product Description 78
Vaginal Ring – HIV Protection – Product Status 78
Vaginal Ring – HIV Protection – Product Description 79
Covidrops LLP Pipeline Products & Ongoing Clinical Trials Overview 80
Nasal Spray – COVID-19 – Product Status 80
Nasal Spray – COVID-19 – Product Description 80
Dare Bioscience Inc Pipeline Products & Ongoing Clinical Trials Overview 81
DARE-FRT1 Intravaginal Ring – Product Status 81
DARE-FRT1 Intravaginal Ring – Product Description 81
DARE-HRT1 Intravaginal Ring – Product Status 82
DARE-HRT1 Intravaginal Ring – Product Description 82
DARE-OAB1 – Product Status 82
DARE-OAB1 – Product Description 83
DARE-VVA1 Intravaginal Ring – Product Status 83
DARE-VVA1 Intravaginal Ring – Product Description 83
Dare Bioscience Inc – Ongoing Clinical Trials Overview 84
DARE-HRT1 Intravaginal Ring – A Phase 1/2, Open-label, Parallel Group Study to Evaluate the Safety and Pharmacokinetics of DARE-HRT1 (80ug Estradiol/4 Mg Progesterone and 160ug Estradiol/8 Mg Progesterone Intravaginal Rings) Over 12 Weeks in Healthy Postmenopausal Women 85
Eledon Pharmaceuticals Inc Pipeline Products & Ongoing Clinical Trials Overview 86
OP0201 – Product Status 86
OP0201 – Product Description 86
Emergent BioSolutions Inc Pipeline Products & Ongoing Clinical Trials Overview 87
SIAN – Product Status 87
SIAN – Product Description 87
Emergent BioSolutions Inc – Ongoing Clinical Trials Overview 88
SIAN – Phase 1, Open Label, Single Ascending Dose Study to Evaluate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of Stabilized Isoamyl Nitrite (SIAN) Nasal Spray in Healthy Adults 89
EmphyCorp Inc Pipeline Products & Ongoing Clinical Trials Overview 90
N115 Non-Steroidal Nasal Spray – Product Status 90
N115 Non-Steroidal Nasal Spray – Product Description 90
Ena Respiratory Pty Ltd Pipeline Products & Ongoing Clinical Trials Overview 91
INNA-051 Nasal Spray – Product Status 91
INNA-051 Nasal Spray – Product Description 91
Ena Respiratory Pty Ltd – Ongoing Clinical Trials Overview 92
INNA-051 Nasal Spray – A Randomized, Double Blind, Placebo-controlled Study to Evaluate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of Single and Multiple Ascending, Intranasal INNA-051 Doses in Healthy Adults 93
Eureka Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 94
InvisiMask Nasal Spray – Product Status 94
InvisiMask Nasal Spray – Product Description 94
Eveon SAS Pipeline Products & Ongoing Clinical Trials Overview 95
Dropper System – Product Status 95
Dropper System – Product Description 95
Evestra Inc Pipeline Products & Ongoing Clinical Trials Overview 96
EVE112 – Vaginal Ring – Product Status 96
EVE112 – Vaginal Ring – Product Description 96
EVE116 – Vaginal Ring – Product Status 97
EVE116 – Vaginal Ring – Product Description 97
EVE120 – Vaginal Ring – Product Status 97
EVE120 – Vaginal Ring – Product Description 98
EVE122 – Vaginal Ring – Product Status 98
EVE122 – Vaginal Ring – Product Description 98
Vaginal Ring For Preterm Labor – Product Status 99
Vaginal Ring For Preterm Labor – Product Description 99
Evofem Biosciences Inc Pipeline Products & Ongoing Clinical Trials Overview 100
Amphora/QGriffithsin Fast-Dissolving Vaginal Insert – Product Status 100
Amphora/QGriffithsin Fast-Dissolving Vaginal Insert – Product Description 101
Ferring Pharmaceuticals AS Pipeline Products & Ongoing Clinical Trials Overview 102
C-Vad Vaginal Insert – Product Status 102
C-Vad Vaginal Insert – Product Description 102
Premis Vaginal Insert – Product Status 103
Premis Vaginal Insert – Product Description 103
Firebrick Pharma Pty Ltd Pipeline Products & Ongoing Clinical Trials Overview 104
Nasodine Nasal Spray – Adult – Product Status 104
Nasodine Nasal Spray – Adult – Product Description 104
Nasodine Nasal Spray – Junior – Product Status 105
Nasodine Nasal Spray – Junior – Product Description 105
Firebrick Pharma Pty Ltd – Ongoing Clinical Trials Overview 106
Nasodine Nasal Spray – Adult – Confirmatory Phase III study of Nasodine Nasal Spray (Povidone-iodine 0.5%) as a Symptomatic Treatment for Early Stage Common Cold in the Natural Setting 107
Nasodine Nasal Spray – Adult – Reduction of Nasal Shedding of SARS-CoV-2 in COVID-19 Positive Patients by the Use of Nasodine (Povidone-iodine 0.5%) Nasal Spray 107
GeneOne Life Science Inc Pipeline Products & Ongoing Clinical Trials Overview 108
GLS-1200 Nasal Spray – Product Status 108
GLS-1200 Nasal Spray – Product Description 108
Ghent University Pipeline Products & Ongoing Clinical Trials Overview 109
Vaginal DL-Lactic Acid Releasing Ring – Product Status 109
Vaginal DL-Lactic Acid Releasing Ring – Product Description 109
Glenmark Pharmaceuticals Ltd Pipeline Products & Ongoing Clinical Trials Overview 110
Ryaltris Nasal Spray – Product Status 111
Ryaltris Nasal Spray – Product Description 111
Glenmark Pharmaceuticals Ltd – Ongoing Clinical Trials Overview 112
Ryaltris Nasal Spray – A Phase III Clinical Study to Evaluate the Efficacy, Safety and Pharmacokinetics of Ryaltris Compound Nasal Spray for the Treatment of Allergic Rhinitis or Rhinoconjunctivitis Symptoms in Patients 113
Grasses of Eden Ltd Pipeline Products & Ongoing Clinical Trials Overview 114
Nasal Spray Device – Product Status 114
Nasal Spray Device – Product Description 114
Grasses of Eden Ltd – Ongoing Clinical Trials Overview 115
Nasal Spray Device – A Randomized, Double Blind, Placebo-controlled Study to Evaluate the Efficacy of Lavandula Angustifolia Containing Nasal Spray Medical Device in Preventing Deterioration of COVID-19 in Symptomatic Patients 116
Grunenthal Ltd Pipeline Products & Ongoing Clinical Trials Overview 117
Estradiol + Progesterone Vaginal Ring – Product Status 117
Estradiol + Progesterone Vaginal Ring – Product Description 117
Fertiring – Product Status 118
Fertiring – Product Description 118
Halberd Corp Pipeline Products & Ongoing Clinical Trials Overview 119
Halberd Nasal Spray – Product Status 119
Halberd Nasal Spray – Product Description 119
Harm Reduction Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 120
Rivive – Product Status 120
Rivive – Product Description 120
Hebrew University of Jerusalem Pipeline Products & Ongoing Clinical Trials Overview 121
Buccal Drug Delivery Device – Oral Cavity Disorders – Product Status 121
Buccal Drug Delivery Device – Oral Cavity Disorders – Product Description 121
Hikma Pharmaceuticals Plc Pipeline Products & Ongoing Clinical Trials Overview 122
Buprenorphine/Naloxone Sublingual Spray – Product Status 122
Buprenorphine/Naloxone Sublingual Spray – Product Description 122
EpiPen – Product Status 123
EpiPen – Product Description 123
Imbed Biosciences Inc Pipeline Products & Ongoing Clinical Trials Overview 124
Nasal Spray – Product Status 124
Nasal Spray – Product Description 124
Institute for Research and Innovation in Health Pipeline Products & Ongoing Clinical Trials Overview 125
TFV/EFV Nanoparticle Vaginal Delivery System – Product Status 125
TFV/EFV Nanoparticle Vaginal Delivery System – Product Description 125
Insys Therapeutics Inc (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 126
Buprenorphine Sublingual Spray Device – Product Status 126
Buprenorphine Sublingual Spray Device – Product Description 126
Naloxone Sublingual Spray – Product Status 127
Naloxone Sublingual Spray – Product Description 127
Ondansetron Sublingual Spray – Product Status 127
Ondansetron Sublingual Spray – Product Description 127
IntelGenx Corp Pipeline Products & Ongoing Clinical Trials Overview 128
Montelukast VersaFilm – Product Status 128
Montelukast VersaFilm – Product Description 128
IntelGenx Corp – Ongoing Clinical Trials Overview 129
Montelukast VersaFilm – A Multicenter, Randomized, Double-blind, Placebo-controlled Clinical Study to Evaluate the Use of Montelukast Versafilm for the Treatment of Parkinson’s Disease (PD) 130
Montelukast VersaFilm – A Randomized Phase IIa, Multi-center, Double-blind, Placebo-controlled Study to Assess the Safety, Feasibility, Tolerability, and Efficacy of a New Buccal Film of Montelukast in Patients with Mild to Moderate Alzheimer’s Disease 130
International Partnership For Microbicides Pipeline Products & Ongoing Clinical Trials Overview 131
Dapivirine-Contraceptive Ring – Product Status 131
Dapivirine-Contraceptive Ring – Product Description 131
International Partnership For Microbicides – Ongoing Clinical Trials Overview 132
Dapivirine-Contraceptive Ring – A Phase I, Open-label, Randomized, Crossover Trial to Investigate the Relative Bioavailability of the 25 Mg Dapivirine Vaginal Ring 004 Inserted Every 30 Days and the 100 Mg Dapivirine Vaginal Ring 008 Inserted for 90 Days in Healthy Female Participants 133
Dapivirine-Contraceptive Ring – A Randomized, Double-Blind, Phase 1b Study in Healthy HIV-Negative Women to Evaluate the Pharmacokinetics, Safety, and Bleeding Patterns Associated With 90-Day Use of Core-Sheath Vaginal Rings Releasing Dapivirine and Levonorgestrel 133
ITC Life Sciences and Technology Centre Pipeline Products & Ongoing Clinical Trials Overview 134
Savlon Nasal Spray – Product Status 134
Savlon Nasal Spray – Product Description 134
ITF Pharma Inc Pipeline Products & Ongoing Clinical Trials Overview 135
Progesterone Vaginal Ring – Luteal Support – Product Status 135
Progesterone Vaginal Ring – Luteal Support – Product Description 135
Janssen Sciences Ireland UC Pipeline Products & Ongoing Clinical Trials Overview 136
Dapivirine Ring – Product Status 136
Dapivirine Ring – Product Description 136
Janssen Sciences Ireland UC – Ongoing Clinical Trials Overview 137
Dapivirine Ring – Phase 3b, Randomized, Open Label Safety Trial of Dapivirine Vaginal Ring and Oral TRUVADA Use in Pregnancy 138
Kane Biotech Inc Pipeline Products & Ongoing Clinical Trials Overview 139
Nasal Spray – Chronic Sinusitis – Product Status 139
Nasal Spray – Chronic Sinusitis – Product Description 139
Karessa Pharma AB Pipeline Products & Ongoing Clinical Trials Overview 140
K-01 – Product Status 140
K-01 – Product Description 140
K-02 – Product Status 141
K-02 – Product Description 141
K-03 – Product Status 141
K-03 – Product Description 142
K-21 – Product Status 142
K-21 – Product Description 142
Karp Lab Pipeline Products & Ongoing Clinical Trials Overview 143
Nasal Spray – SARS-CoV-2 – Product Status 143
Nasal Spray – SARS-CoV-2 – Product Description 143
Kiora Pharmaceuticals Inc Pipeline Products & Ongoing Clinical Trials Overview 144
EyeGate II Delivery System – Product Status 144
EyeGate II Delivery System – Product Description 145
Kytogenics Pharmaceuticals Inc Pipeline Products & Ongoing Clinical Trials Overview 146
Drug Delivery Device – Intestinal Cystitis – Product Status 146
Drug Delivery Device – Intestinal Cystitis – Product Description 146
Laboratoire HRA Pharma SAS Pipeline Products & Ongoing Clinical Trials Overview 147
UPA – CVR – Product Status 147
UPA – CVR – Product Description 147
Lachesis Biosciences Ltd Pipeline Products & Ongoing Clinical Trials Overview 148
Rivamist Nasal Spray – Product Status 148
Rivamist Nasal Spray – Product Description 148
Leyden Laboratories BV Pipeline Products & Ongoing Clinical Trials Overview 149
Liquid Nasal Spray – Product Status 149
Liquid Nasal Spray – Product Description 149
Lobe Sciences Ltd Pipeline Products & Ongoing Clinical Trials Overview 150
Nasal Mist Device – Product Status 150
Nasal Mist Device – Product Description 150
Lyra Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 151
LYR-210 Transmucosal Delivery System – Product Status 151
LYR-210 Transmucosal Delivery System – Product Description 151
LYR-220 – Product Status 152
LYR-220 – Product Description 152
Lyra Therapeutics Inc – Ongoing Clinical Trials Overview 153
LYR-210 Transmucosal Delivery System – ENLIGHTEN 1: A Phase III, Randomized, Blinded, Controlled, Parallel-Group Trial to Evaluate the Efficacy and Safety of LYR-210 for the Treatment of Chronic Rhinosinusitis (CRS) in Adults 154
LYR-210 Transmucosal Delivery System – ENLIGHTEN 2: A Phase III, Randomized, Blinded, Controlled, Parallel-Group Trial to Evaluate the Efficacy and Safety of LYR-210 for the Treatment of Chronic Rhinosinusitis in Adults 154
LYR-220 – BEACON: A Phase II, Patient-blinded, Two-part, Randomized, Parallel-group Trial to Evaluate the Safety, Tolerability, Pharmacokinetics, and Efficacy of LYR-220 in Chronic Rhinosinusitis (CRS) Patients Who Have Had a Prior Ethmoidectomy 155
Magee-Womens Research Institute Pipeline Products & Ongoing Clinical Trials Overview 156
EFdA-P+Progestin Intravaginal Film – Product Status 156
EFdA-P+Progestin Intravaginal Film – Product Description 156
MassBiologics Pipeline Products & Ongoing Clinical Trials Overview 157
mAb 2C7+TDF IVR – Product Status 157
mAb 2C7+TDF IVR – Product Description 157
McNeil AB Pipeline Products & Ongoing Clinical Trials Overview 158
Antiviral Polymeric Nasal Spray – Product Status 158
Antiviral Polymeric Nasal Spray – Product Description 158
Medlab Clinical Ltd Pipeline Products & Ongoing Clinical Trials Overview 159
NanaBis – Product Status 159
NanaBis – Product Description 159
Medlab Clinical Ltd – Ongoing Clinical Trials Overview 160
NanaBis – An Observational Study Investigating and Auditing the Safety, Tolerability and Further Efficacy Characteristics of a Pharmaceutical Grade Cannabis Medicine (NanaBis) Prescribed to Eligible Patients for the Management of Cancer Related or Non-cancer Related Pain in General and Specialty Medical Practices 161
Merck & Co Inc Pipeline Products & Ongoing Clinical Trials Overview 162
ENG-CVR – Product Status 162
ENG-CVR – Product Description 162
Merck Serono SA Pipeline Products & Ongoing Clinical Trials Overview 163
MK-8342 Medicated IUS – Product Status 163
MK-8342 Medicated IUS – Product Description 163
Mucommune LLC Pipeline Products & Ongoing Clinical Trials Overview 164
HCA+VRC01+N6 IVR – Product Status 164
HCA+VRC01+N6 IVR – Product Description 164
LA-Releasing Intravaginal Ring – Product Status 165
LA-Releasing Intravaginal Ring – Product Description 165
Non-Hormonal Contraceptive Intravaginal Ring – Product Status 165
Non-Hormonal Contraceptive Intravaginal Ring – Product Description 166
Mycrodose Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 167
Sublingual Drug Delivery System – LSD – Product Status 167
Sublingual Drug Delivery System – LSD – Product Description 167
Mystic Pharmaceuticals Inc Pipeline Products & Ongoing Clinical Trials Overview 168
MystiFen – Product Status 168
MystiFen – Product Description 168
MystiZof – Product Status 169
MystiZof – Product Description 169
Nanosys Inc Pipeline Products & Ongoing Clinical Trials Overview 170
Gecko-Inspired Drug Delivery Device – Product Status 170
Gecko-Inspired Drug Delivery Device – Product Description 170
Nasus Medical Inc Pipeline Products & Ongoing Clinical Trials Overview 171
Catheter-Based Atomization Drug Delivery Device – Product Status 171
Catheter-Based Atomization Drug Delivery Device – Product Description 171
Nasus Pharma Ltd Pipeline Products & Ongoing Clinical Trials Overview 172
FMXIN001 Naloxone Powder-Based Nasal Spray – Product Status 172
FMXIN001 Naloxone Powder-Based Nasal Spray – Product Description 172
Nemera La Verpilliere Pipeline Products & Ongoing Clinical Trials Overview 173
Electronic Nasal Device – Product Status 173
Electronic Nasal Device – Product Description 173
NorInvent AB Pipeline Products & Ongoing Clinical Trials Overview 174
XtriG Drug Delivery Device – Product Status 174
XtriG Drug Delivery Device – Product Description 174
Novlead Biotechnology Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 175
iNOfirst Antiviral Spray – Product Status 175
iNOfirst Antiviral Spray – Product Description 175
Olfax LLC Pipeline Products & Ongoing Clinical Trials Overview 176
Intranasal Delivery Device – Product Status 176
Intranasal Delivery Device – Product Description 176
Opiant Pharmaceuticals Inc Pipeline Products & Ongoing Clinical Trials Overview 177
OPNT001 Opioid Antagonist Nasal Spray – Product Status 177
OPNT001 Opioid Antagonist Nasal Spray – Product Description 177
OPNT002 Opioid Antagonist Nasal Spray – Product Status 178
OPNT002 Opioid Antagonist Nasal Spray – Product Description 178
Opiant Pharmaceuticals Inc – Ongoing Clinical Trials Overview 179
OPNT002 Opioid Antagonist Nasal Spray – Randomised, Double-blind, Placebo-controlled Trial Evaluating the Effects of Naltrexone Hydrochloride Nasal Spray on Alcohol Consumption in Alcohol Usse Disorder 180
OptiNose AS Pipeline Products & Ongoing Clinical Trials Overview 181
OptiNose Bi-Directional Breath Powered Orexin-A Delivery Device – Product Status 181
OptiNose Bi-Directional Breath Powered Orexin-A Delivery Device – Product Description 181
XHANCE – Product Status 182
XHANCE – Product Description 182
OptiNose AS – Ongoing Clinical Trials Overview 183
XHANCE – 16-Week Randomized Double-blind Placebo Controlled Parallel-Group Multicenter Study Evaluating the Efficacy and Safety of OPN-375 186 µg Twice a Day in Adolescents with Bilateral Nasal Polyps Followed with 12-Week Open-label Treatment Phase 184
OptiNose Inc Pipeline Products & Ongoing Clinical Trials Overview 185
Optinose Exhalation Delivery Device – Product Status 185
Optinose Exhalation Delivery Device – Product Description 185
Organon Pharma (UK) Ltd Pipeline Products & Ongoing Clinical Trials Overview 186
Etonogestrel + 17ß-Estradiol Vaginal Ring – Product Status 186
Etonogestrel + 17ß-Estradiol Vaginal Ring – Product Description 186
Orion Biotechnology Canada Ltd Pipeline Products & Ongoing Clinical Trials Overview 187
OB-002 IVR – Product Status 187
OB-002 IVR – Product Description 187
Orphanse Healthcare SAS Pipeline Products & Ongoing Clinical Trials Overview 188
ORH01 Drug Delivery Device – Product Status 188
ORH01 Drug Delivery Device – Product Description 188
Oyster Point Pharma Inc Pipeline Products & Ongoing Clinical Trials Overview 189
Tyrvaya Nasal Spray – Product Status 189
Tyrvaya Nasal Spray – Product Description 189
P&B Group Pipeline Products & Ongoing Clinical Trials Overview 190
ADW S-100 Ionized Water Nasal Spray – Product Status 190
ADW S-100 Ionized Water Nasal Spray – Product Description 190
P&B Group – Ongoing Clinical Trials Overview 191
ADW S-100 Ionized Water Nasal Spray – Efficacy of ADW S-100 Ionized Water Nasal Spray in Decreasing Symptoms of Allergic Rhinitis: Randomized, Double-blind, Placebo-controlled Trial 192
Palette Life Sciences AB Pipeline Products & Ongoing Clinical Trials Overview 193
Lidbree – Product Status 193
Lidbree – Product Description 193
Parnell Pharmaceuticals Inc Pipeline Products & Ongoing Clinical Trials Overview 194
Nomovid Nasal Spray – Product Status 194
Nomovid Nasal Spray – Product Description 194
Phoenix Life Sciences International Ltd Pipeline Products & Ongoing Clinical Trials Overview 195
Phoenix Neuro – Product Status 195
Phoenix Neuro – Product Description 195
Phoenix Pain – Product Status 196
Phoenix Pain – Product Description 196
PHOENIX Sport Plus – Product Status 196
PHOENIX Sport Plus – Product Description 197
pHOXBIO Ltd Pipeline Products & Ongoing Clinical Trials Overview 198
pHOXWELL – Product Status 198
pHOXWELL – Product Description 198
pHOXBIO Ltd – Ongoing Clinical Trials Overview 199
pHOXWELL – A Phase I Study to Evaluate the Safety of pHOXWELL Nasal Spray in the Prevention of SARS-CoV-2 Infection 200
Pocket Naloxone Corp Pipeline Products & Ongoing Clinical Trials Overview 201
Intranasal Spray – Naloxone – Product Status 201
Intranasal Spray – Naloxone – Product Description 201
Polyrizon Ltd Pipeline Products & Ongoing Clinical Trials Overview 202
Intranasal Delivery System – Product Status 202
Intranasal Delivery System – Product Description 202
RDD Pharma Ltd (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 203
Capository – Product Status 203
Capository – Product Description 203
Receptor Life Sciences Inc Pipeline Products & Ongoing Clinical Trials Overview 204
RLS103 – Product Status 204
RLS103 – Product Description 204
Renown Pharmaceuticals Pvt Ltd Pipeline Products & Ongoing Clinical Trials Overview 205
Sub-Lingual Apomorphine Spray – Product Status 205
Sub-Lingual Apomorphine Spray – Product Description 205
Rhinomed Ltd Pipeline Products & Ongoing Clinical Trials Overview 206
Intranasal Drug Delivery Device – Acute Migraine – Product Status 206
Intranasal Drug Delivery Device – Acute Migraine – Product Description 206
Sanotize Research and Development Corp Pipeline Products & Ongoing Clinical Trials Overview 207
Fabispray – Product Status 207
Fabispray – Product Description 207
Sanotize Research and Development Corp – Ongoing Clinical Trials Overview 208
Fabispray – A Double-blinded, Placebo-controlled Parallel, Phase 3 Clinical Efficacy Study Evaluating Nitric Oxide Nasal Spray (NONS) to Treat and Prevent the Exacerbation of Infection in Individuals with Documented Asymptomatic or Mild COVID-19 209
Fabispray – A Phase III Study of SaNOtize Nitric Oxide Nasal Spray for the Early Treatment of Patients with COVID-19 and Its Variants 209
Scientus Pharma Inc Pipeline Products & Ongoing Clinical Trials Overview 210
RapidMist Buccal Delivery System – Product Status 210
RapidMist Buccal Delivery System – Product Description 210
Seelos Therapeutics, Inc. Pipeline Products & Ongoing Clinical Trials Overview 211
SLS-002 Intranasal Device – Product Status 211
SLS-002 Intranasal Device – Product Description 211
Seelos Therapeutics, Inc. – Ongoing Clinical Trials Overview 212
SLS-002 Intranasal Device – A 2-part Phase 2 Study to Assess the Efficacy, Safety, and Tolerability of SLS-002 (Intranasal Racemic Ketamine) Administered to Adults with Major Depressive Disorder at Imminent Risk of Suicide 213
Shin Nippon Biomedical Laboratories Ltd Pipeline Products & Ongoing Clinical Trials Overview 214
µco System – Product Status 214
µco System – Product Description 214
SipNose Ltd Pipeline Products & Ongoing Clinical Trials Overview 215
Cribriform Targeted Device – Product Status 215
Cribriform Targeted Device – Product Description 216
South Valley University Pipeline Products & Ongoing Clinical Trials Overview 217
Ivermectin Nasal Spray – Product Status 217
Ivermectin Nasal Spray – Product Description 217
Starpharma Holdings Ltd Pipeline Products & Ongoing Clinical Trials Overview 218
Viraleze Nasal Spray – Product Status 218
Viraleze Nasal Spray – Product Description 218
The Population Council Inc Pipeline Products & Ongoing Clinical Trials Overview 219
EEQ Intravaginal Ring – Product Status 219
EEQ Intravaginal Ring – Product Description 219
Therakind Ltd Pipeline Products & Ongoing Clinical Trials Overview 220
DriDose – Product Status 220
DriDose – Product Description 220
Union Therapeutics AS Pipeline Products & Ongoing Clinical Trials Overview 221
Niclosamide Nasal Spray – Product Status 221
Niclosamide Nasal Spray – Product Description 221
University of California Davis Pipeline Products & Ongoing Clinical Trials Overview 222
Copper Intravaginal Ring (Cu-IVR) – Product Status 222
Copper Intravaginal Ring (Cu-IVR) – Product Description 222
University of Erlangen-Nurnberg Pipeline Products & Ongoing Clinical Trials Overview 223
Carragelose Nasal Spray – Product Status 223
Carragelose Nasal Spray – Product Description 223
University of Kentucky Pipeline Products & Ongoing Clinical Trials Overview 224
Povidone-Iodine Nasal Spray – Product Status 224
Povidone-Iodine Nasal Spray – Product Description 224
University of Kentucky – Ongoing Clinical Trials Overview 225
Povidone-Iodine Nasal Spray – Povidone-Iodine Intranasal for Prophylaxis in Front-line Health-care Personnel and Inpatients During the Sars-CoV-2 Pandemic 226
University of Louisville Pipeline Products & Ongoing Clinical Trials Overview 227
Q-Griffithsin Nasal Spray – Product Status 227
Q-Griffithsin Nasal Spray – Product Description 227
University of Melbourne Pipeline Products & Ongoing Clinical Trials Overview 228
At-Home Nasal Spray – COVID-19 – Product Status 228
At-Home Nasal Spray – COVID-19 – Product Description 228
University of Melbourne – Ongoing Clinical Trials Overview 229
At-Home Nasal Spray – COVID-19 – Study to Evaluate the Efficacy of At-Home Nasal Spray – COVID-19 in Patients with COVID-19 230
University of North Carolina at Chapel Hill Pipeline Products & Ongoing Clinical Trials Overview 231
Dapivirine + Pritelivir + Levonorgestrel 3D Printed IVR – Product Status 231
Dapivirine + Pritelivir + Levonorgestrel 3D Printed IVR – Product Description 231
Islatravir+Etonogestrel/Ethinyl Estradiol 3D Printed IVR – Product Status 232
Islatravir+Etonogestrel/Ethinyl Estradiol 3D Printed IVR – Product Description 232
University of Pittsburgh Pipeline Products & Ongoing Clinical Trials Overview 233
HerShield – Product Status 233
HerShield – Product Description 233
University of South Florida Pipeline Products & Ongoing Clinical Trials Overview 234
Drug Delivery Device – Ovarian Cancer – Product Status 234
Drug Delivery Device – Ovarian Cancer – Product Description 234
University of Texas at Austin Pipeline Products & Ongoing Clinical Trials Overview 235
Transmucosal Patch – Product Status 235
Transmucosal Patch – Product Description 235
University of the Witwatersrand Pipeline Products & Ongoing Clinical Trials Overview 236
Vagitab – Product Status 236
Vagitab – Product Description 236
University of Waterloo Pipeline Products & Ongoing Clinical Trials Overview 237
HIV Prevention Device – Product Status 237
HIV Prevention Device – Product Description 237
Virpax Pharmaceuticals Inc Pipeline Products & Ongoing Clinical Trials Overview 238
AnQlar – Product Status 238
AnQlar – Product Description 238
MMS019 – Product Status 239
MMS019 – Product Description 239
VistaGen Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 240
PH10 Nasa
![]()